Local Passive Warming Administered During the Transition Phase Impairs Subsequent Isokinetic Exercise Performance
Article information
Trans Abstract
PURPOSE
Performing a warm-up prior to a sports competition is essential for preventing injuries and maximizing subsequent performance. Passive warming has therefore received attention because it can effectively maintain an elevated muscle temperature (T m) after a warm-up. The present study aimed to investigate the effects of local passive warming (LPW) administered during the transition phase between active warm-up and isokinetic exercises.
METHODS
Eleven healthy men participated in this randomized crossover study. During the 30-minute transition period, a customized water-perfused warming pad was placed on the anterior thighs of the dominant legs (+3°C or +6°C based on the baseline skin temperature) or no intervention for the control group (CON). Subsequently, the total work, peak torque, and average power during isokinetic knee extensions (10 repetitions at a speed of 60°/sce) were measured. Using surface electromyography (EMG), the root mean square and mean frequency from the rectus femoris during knee extension were measured to assess neuromuscular activity.
RESULTS
Total work (+6°C 1,289.8±389.3 vs. CON 1,437.3±408.2 Nm, p =.042), peak torque (+6°C 146.0±33.3 vs. CON 162.9±37.9 Nm, p=.022), and average power (+6°C 77.7±19.8 vs. CON 90.2±25.3 W, p=.012) were lower in the LPW +6°C group than in the CON group. EMG RMS was lower in the LPW +6°C group than in the CON group (+6°C 58.4±22.3 vs. CON 78.6±33.0 % MVC, p =.005). MF was higher in the LPW +6°C group than in the CON group (+6°C 87.5±12.4 vs. CON 81.4±11.0 Hz, p=.016).
CONCLUSIONS
Local passive warming administered during the transition phase was not an appropriate strategy for boosting the performance of subsequent isokinetic exercises. Accordingly, the application of passive warming should be carefully chosen before exercise commences.
INTRODUCTION
Performing a warm-up prior to a sports competition is essential to the prevention of injuries and maximizes subsequent exercise performance [1–3]. Warm-up induced elevations in muscle temperature (T m) can increase muscular contraction speed [1,4], nerve conduction velocity [1], and boost metabolism [5–7], consequently leading to improvements in exercise performance. According to prior studies, active warm-up exercise can increase T m by 2.5-4°C [2,8], with each 1°C rise in T m enhancing muscle power output by 4-10% [3,5,8]. For this reason, researchers have attempted to optimize warm-up exercise components, such as intensity and duration, in an effort to maximize athletic performance [9,10].
In many sports, however, the physiological benefits gained from engaging in a prior warm-up exercise are, at least in part, lost during the transition phase (i.e., the period between completion of the warm-up and the start of the event) because T m declines immediately upon exercise cessation [10–12]. A 1°C decrease in T m leads to a 3% decrease in muscle strength [3], and it is not uncommon for athletes to experience a transition period in excess of 30 minutes [6]. Studies have shown that exercise performance is reduced by 15-20% following a 20-40 minutes transition period [13,14]. It would therefore be helpful to utilize a means of maintaining an elevated T m throughout the transition period [9,10,15].
Passive warming has attracted interest as a possible means of achieving this goal, as it can effectively maintain an elevated T m after a warm-up exercise, but does not result in the depletion of energy substrates [6,11]. Various passive warming modalities have been investigated, including hot water immersion [3], heating pads [5], blizzard survival jack-ets [11], heated pants [12], and passive heat retention garments [16]. However, passive warming has shown inconsistent results in terms of measured exercise performance. Passive warming used during the transition phase has been shown to improve exercise performance of swimming [4], cycling [8,12], sprinting [17], and vertical jumping [18], but have no beneficial effects on repeated sprints [19] or recovery after resistance training [20]. Furthermore, it has even been shown to have a negative effect on the performance of repetitive knee extensor exercises (time to exhaustion) [21] and muscular endurance during isometric knee extension [22,23]. To better understand discrepancies regarding the performance effect of passive warming intervention, contributing factors such as warming modalities and type of exercise need to be investigated. Then, passive warming can be relied upon as an effective pre-conditioning option for athletes and coaches.
It is well known that an increased T m leads to elevated muscle contraction speed and rate of force development [15,24,25], which could result in improvement in explosive exercise performance [8,12,17,18] that is dominantly influenced by contraction speed. However, it remains unan-swered whether muscle strength (i.e., peak and average torque) is affected by increased T m during muscular contraction. Using an isokinetic dynamometer that enables to assess muscle strength throughout the range of motion with muscular contraction speed fixed, this study therefore aims to investigate the effects of local passive warming administered during the transition phase on muscular strength during repeated isokinetic knee extensions. Based on previous studies showing blunted isometric knee extension performance following an increased T m [22,23], we hypothesized in the current study that muscle strength during isokinetic knee extensions may be blunted in response to local passive warming.
METHODS
1. Ethical Approval
The institutional review board (IRB#: JBNU 2019-09-010-002) approved the experimental protocol. The study procedure was explained in detail to the participants. Subjects were verbally informed about experimental procedures and potential risks prior to obtaining a written consent form. The study conformed to the provisions of the Declaration of Helsinki.
2. Participants
Eleven healthy and young subjects (Age: 27.6±1.0 years, Height: 172.9±3.1 cm, Body Weight: 72.6±5.2 kg, BMI: 24.0±1.0 kg/m²) participated in the present investigation. All subjects were free from cardiovascular, metabolic, or orthopedic diseases. Participants were instructed to maintain their regular diet and physical activity during the experiment and were asked to refrain from high-intensity exercise, and the consumption of alcohol and caffeine during the 24 hours preceding each visit. Participants did not eat in the 2 hours preceding the trial. This experiment was conducted in a temperature-controlled laboratory (-22°C and 50% relative humidity).
3. Experimental Design
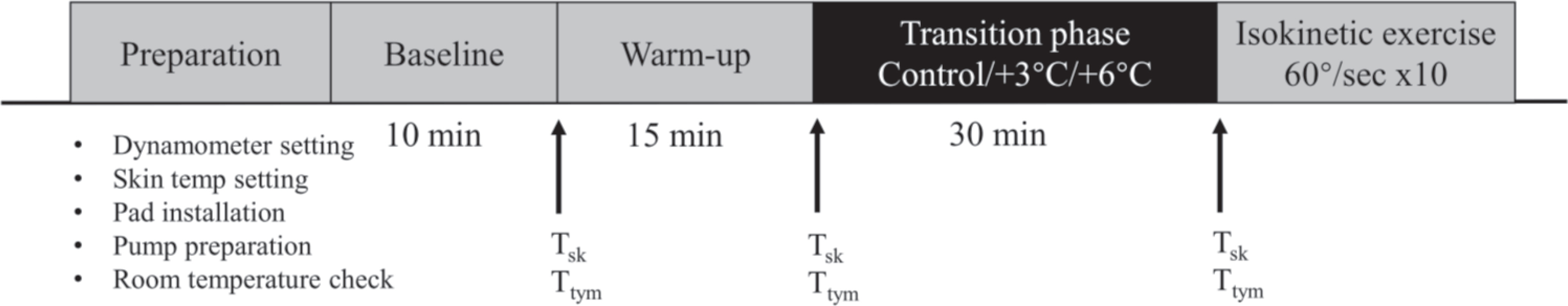
This experiment was a randomized crossover study that included four visits. On the first familiarization visit, all subjects were informed of the experimental process and signed written consent. Their body composition, including their weight and fat-free mass (InBody 720, InBody, Seoul, Korea), were then measured. Each participant was familiarized with the dominant single-leg knee extension exercise (60°/sec, 10 reps) performed on an isokinetic dynamometer (CSMi, Humac Norm, MA, USA). A maximal voluntary contraction (MVC) test with the knee extended at 60 degree was performed to normalize neuromuscular analysis [26]. The three test visits were completed at the same time of day in a randomized order using random sampling generator with at least seven days separation (Fig. 1).
On the test visit, the temperature sensor (moorVMS-LDF2, Moor In-struments, Devon, UK) was placed on one half of the rectus femoris muscle first for baseline skin temperature (T sk) measurement.
A baseline skin temperature (T sk) (moorVMS-LDF2, Moor Instru-ments, UK) was continuously measured during the 10-minute baseline and averaged for last 1 minute as an index of baseline T sk. Tympanic temperature (T tym) (IRT6520, Braun, Kronberg, Germany) was measured three times at the end of the baseline period and averaged for data analysis. Following the 10-minute baseline, subjects performed a 15-minute warm-up exercise consisting of running (8 km/hr for 5-minute), burpee jumps (3×15 reps), and stretching exercise for their lower body. Subjects then sat in an isokinetic device for 30 minutes (i.e., transition phase) with the anterior thigh of their dominant leg covered with a customized water-perfused tube-lined pad (28×34 cm) in which a peristaltic pump (BT100J-1A, Huiyu, Beijing, China) pumped water. During this transition phase, the temperature of the pad was maintained at either thermoneutral T sk (CON), 3°C higher (3°C) than that individual’ s baseline T sk, or 6°C higher (6°C) than that individual’ s baseline T sk. Following the 30-minute transition period, subjects performed ten repetitions of the knee extension exercise on the isokinetic dynamometer at the speed of 60°/sec to assess muscular strength and development of fatigue [27,28].
4. Isokinetic Exercise Performance
Knee extension exercise performance was measured using a dynamometer with Humac Norm software. Before each test, the instrument was calibrated, the input arms were perpendicular and aligned, and the subjects sat in an adjustable chair with safety straps around the waist and across the chest to limit changes in any position during the exercise. The ankle strap on the dynamometer’ s lever arm was placed 2 cm proximal to the lateral malleolus. At the end of the test, an exercise performance report that included measurements of total work (Newton-meter, Nm), peak torque (Newton-Meter, Nm), average power (Watts, W), the joint angle at peak torque (°), and range of motion (°) was produced by the software.
5. Maximal Voluntary Contraction (MVC) Test
The representative maximum EMG signal was evaluated by the performance of an isometric knee extension maximal voluntary contraction. Subjects were placed in a sitting position with the knee extended at 60 degrees from full-length extension and told to forcibly extend their knee against the dynamometer [26]. Subjects were given strong encouragement during the test. MVC EMG was assessed twice, with each test taking 5 seconcds. Only the maximum MVC EMG was recorded and the rest time between contractions was 1 minute.
6. Electromyography (EMG) Measurement and Analysis
The neuromuscular activity was assessed using wireless electromyography (EMG; Tringo EMG Wireless, Delsys Inc., USA) during the knee extension exercise. Before the EMG electrode was attached, hair on the skin surface was scraped, disinfected with a 70% alcohol pad, and thoroughly dried. The EMG sensor was placed at 1/2 on the line from the anterior superior iliac superior to the superior part of the patella (i.e., rectus femoris) based on SENIAM guidelines.
Signals from the rectus femoris were wirelessly recorded with a sampling frequency of 2,000 Hz. Following data collection, mean frequency (MF) and root mean square (RMS) was calculated by analysis software (Delsys EMG Works Analysis 4.2.0, Delsys, MA, USA). The filtering range was set at 10-500 Hz to reduce noise in the low-frequency region and eliminate motion artifacts in the high-frequency region. The root mean square (RMS) was analyzed as % MVC as an indicator of the maximal muscle activation. All EMG amplitude signals as indexed by MF and RMS were normalized to the maximal RMS like previously described [29]. The threshold level of RMS analysis was chosen as 20% on-set and 20% offset, such that raw signals higher than 20% MVC in rising and declining phases based on 100% MVC RMS signals were selected for data analysis, and the selected raw EMG signals were also utilized for MF data analysis [30].
7. Statistical Analysis
Data are presented as mean±SD, and statistical significance was accepted as p <.05. Variables for temperature (T sk and T tym), isokinetic performance for each repetition, and EMG analysis for each repetition were analyzed using a two-way repeated measures ANOVA, followed by Tukey’ s multiple comparisons analysis; the factors were intervention conditions (CON, +3°C and +6°C) and time (stages or repetitions). Group main effects (CON, +3°C and +6°C) in isokinetic performance and EMG analysis for each repetition was separately presented as figs. Absolute standardized effect sizes (Cohen’ s d) were calculated and reported in case of statistical difference between groups. Statistical analyses were performed using GraphPad Prism 9.2.0 software (GraphPad Soft-ware, La Jolla, CA).
RESULTS
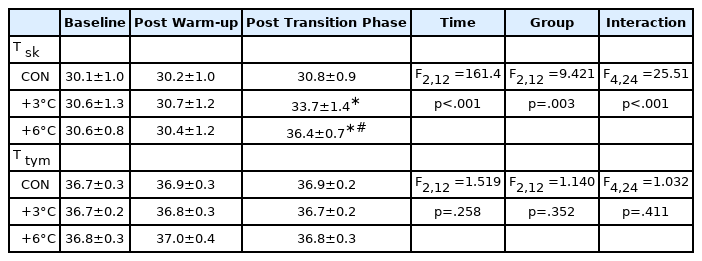
T sk and T tym are presented in Table 1. For T sk, there were significant time (F2,12 =161.4, p <.001), group effects (F2,12 =9.421, p =.003), and interaction (F4,24 =25.51, p <.001). No differences between groups were observed in T sk at baseline (CON 30.1±1.0 vs. +3°C 30.6±1.3°C, p =.479; CON 30.1±1.0 vs. +6°C 30.6±0.8°C, p =.555; +3°C 30.6±1.3 vs. +6°C 30.6±0.8°C, p =.991). T sk was substantially higher compared to CON following the administration of passive warming at +3°C and +6°C (CON 30.8±0.9 vs. +3°C 33.7±1.4, p <.001, ES=2.464) and +6°C (CON 30.8±0.9 vs. +6°C 36.4±0.7°C, p <.001, ES=6.946). For T tym, no significant time (F2,12 =1.519, p =.258), group effects (F2,12 =1.140, p =.352), and interaction (F4,24 =1.032, p =.411) were found. T tym was unchanged in all groups throughout the test (Table 1). The passive warming groups tended to show attenuated total work, peak torque, and average power compared to the CON group (Table 2). For total work, there were significant time (F9,90 =3.128, p =.002) and group effects (F2,20 =3.547, p =.048) but not significant interaction (F18,180 = 1.209, p =.258). Peak torque showed significant group effect (F2,20 =4.389, p =.026) but nonsignificant time effect (F9,90 =1.916, p =.059) and interaction (F18,180 =1.212, p =.255). For average power, there were significant time (F9,90 =2.404, p =.017) and group effects (F2,20 =6.244, p =.008) but not significant interaction (F18,180 =0.8277, p =.666). For total work and peak torque, the +6°C group experienced a more significant reduction at Rep 1 (Total work: +3°C 139.5±39.7 vs. +6°C 121.5±57.8 Nm, p <.001, ES=0.363) and 5 (Peak torque: +3°C 155.5±39.0 vs. +6°C 143.2±27.1 Nm, p =.0002, ES=0.366) relatives to the +3°C group (Table 2).
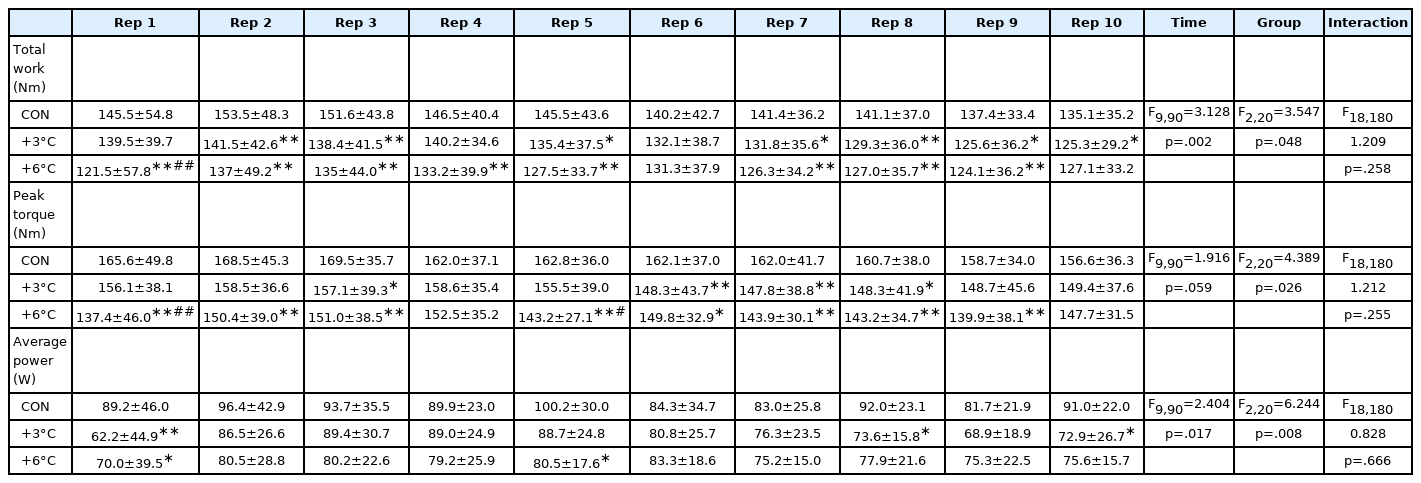
Isokinetic extension performance as total work (TW), peak torque (PT) and average power (AP) per repetition
Group main effects (CON, +3°C and +6°C) in isokinetic performance (Total work, peak torque, and average power) were presented in Fig. 2A-C. There were main intervention effects on total work (F2,20 =3.549, p = .048), peak torque (F2,20 =4.319, p =.028), and average power (F2,20 =6.383, p =.007). Passive warming at +6°C resulted in adverse effects on total work (CON 1437.3±408.2 vs. +6°C 1,289.8±389.3 Nm, p =.042, ES= 0.370) and peak torque (CON 162.9±37.9 vs. +6°C 146.0±33.3 Nm, p = .022, ES=0.474) relative to the CON group while no differences were observed in the +3°C group when compared with the CON group (Total work: CON 1,437.3±408.2 vs. +3°C 1,338.6±354.3 Nm, p =.212; Peak torque: CON 162.9±37.9 vs. +3°C 153.0±38.1 Nm, p =.225). Average power was lower in the passive warming groups (+3°C and +6°C) compared the CON group (CON 90.2±25.3 vs. +3°C 78.7±18.1 W, p =.020, ES=0.523; CON 90.2±25.3 vs. +6°C 77.7±19.8 W, p =.012, ES=0.550).
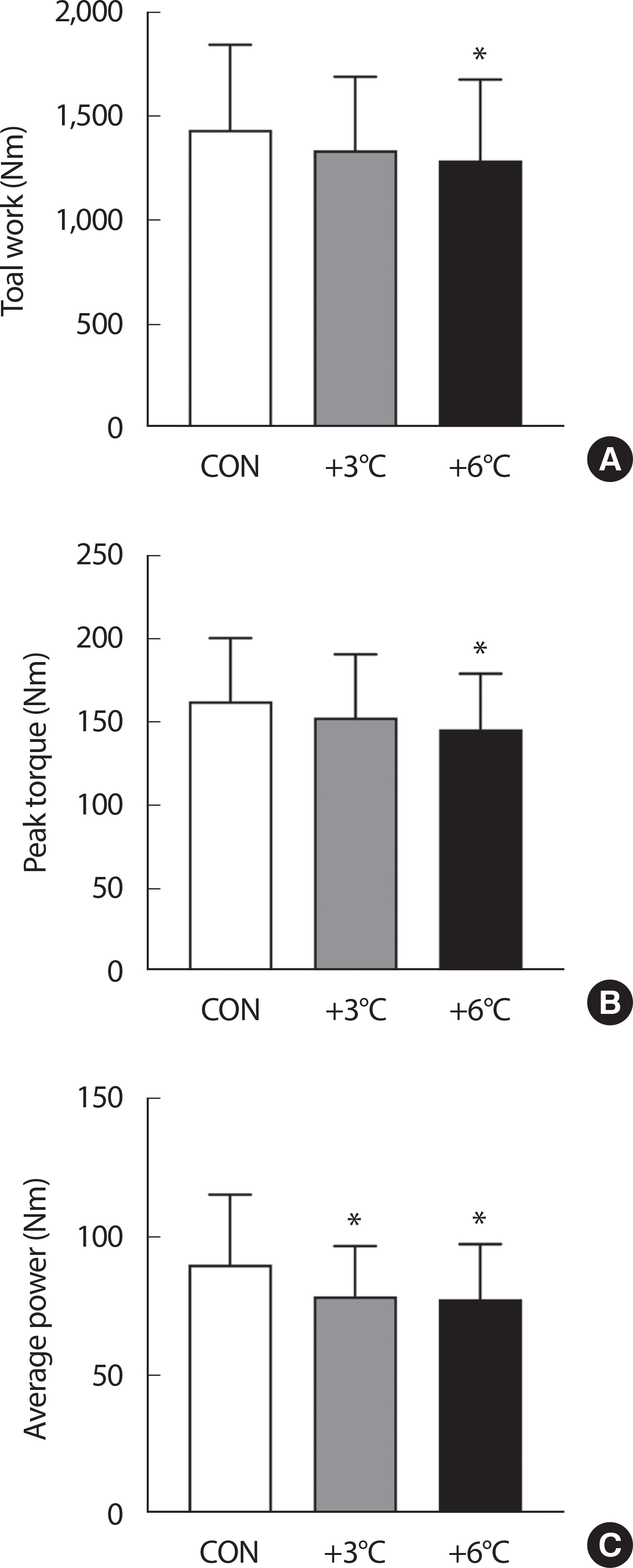
Isokinetic exercise performance. (A) Total work, (B) Peak torque, and (C) average power during a total of ten repetitions of isokinetic knee extensions are shown. n=11, Data are presented as the mean±SD. ∗ p<.05 vs. CON.
Joint angle at peak torque and range of motion during a total of ten repetitions of the isokinetic knee extensions are shown Fig. 3. No differences were detected in the joint angle at peak torque across groups (CON 45.0±13.4 vs. +3°C 46.7±11.2°, p =.891; CON 45.0±13.4 vs. 6°C 52.2±17.2°, p =.163; +3°C 46.7±11.2 vs. +6°C 52.2±17.2°, p =.337). Also, no differences in the range of motion during the isokinetic knee extensions were observed (CON 73.0±14.6 vs. +3°C 73.6±13.4°, p =.991; CON 73.0±14.6 vs. +6°C 76.1±18.4°, p =.802; +3°C 73.6±13.4 vs. +6°C 76.1±18.4°, p =.870).
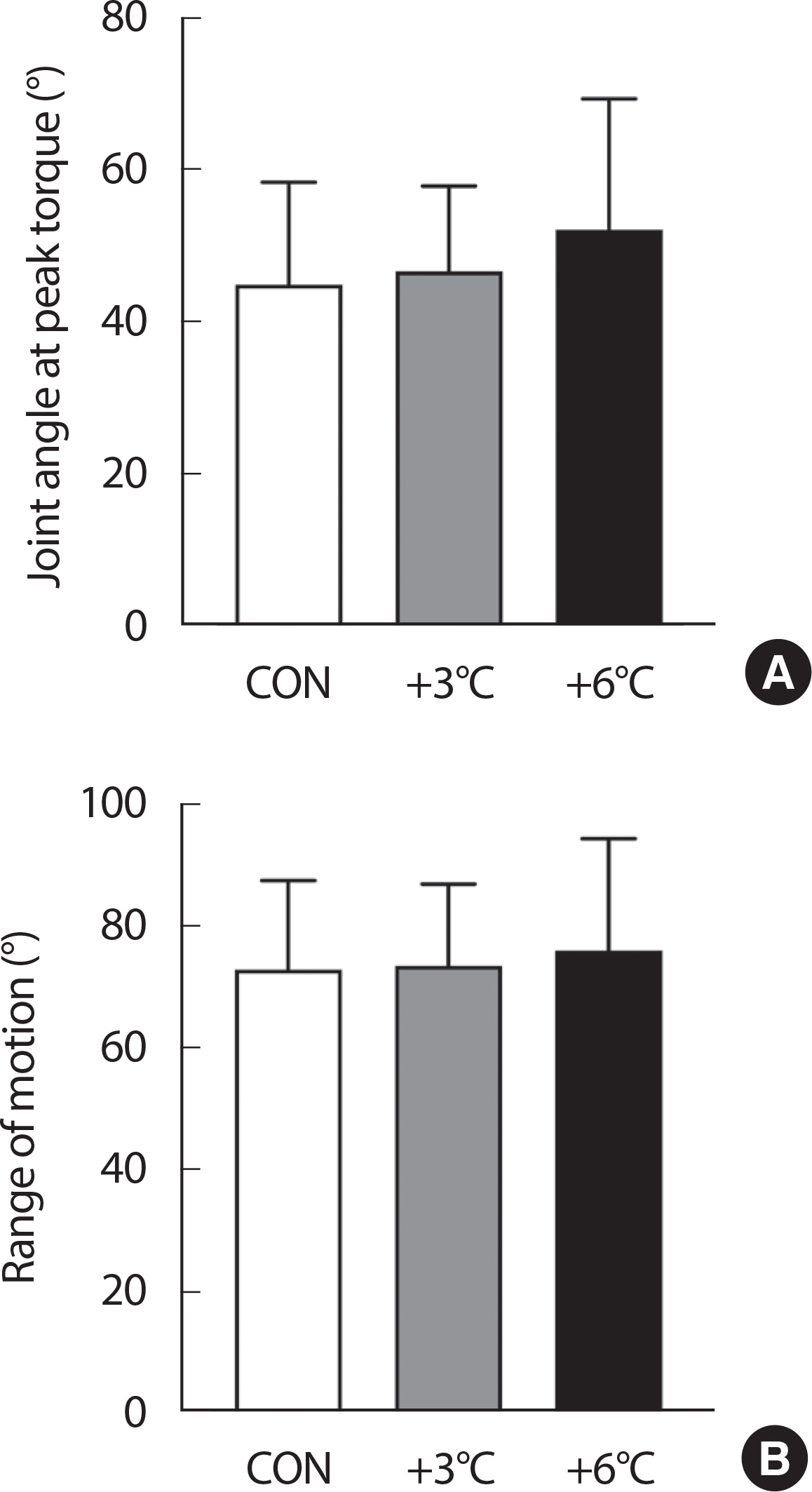
Joint angle at peak torque and range of motion. (A) Joint angle at peak torque and (B) Range of motion during a total of ten repetitions of isokinetic knee extensions are shown. n=11, Data are presented as the mean±SD.
The RMS and MF of each repetition are shown in Table 3. The passive warming groups (+3°C and +6°C) manifested decreased RMS relative to the CON group (Table 3). For RMS, there were not significant time effect (F9,90 =1.460, p =.175) and interaction (F18,180 =1.402, p =.135), and significant group effect (F2,20 =6.614, p =.006). For MF, there were significant time (F9,90 =18.64, p<.001), group effects (F2,20 =4.741, p =.021), and interaction (F18,180 =2.414, p =.002). For RMS, warming at +6°C led to lower at Rep 1 (p =.016, ES=0.265), 3 (p =.033, ES=0.232), 4 (p =.004, ES=0.286), 5 (p =.002, ES=0.385), 7 (p =.003, ES=0.309), 9 (p =.017, ES=0.318), and 10 (p =.005, ES=0.318) than that observed in the +3°C group. MF was higher in both passive warming groups (+3°C and +6°C) than in the CON group. With respect to MF, heating +6°C led to higher at Rep 2 (p=.0003, ES=0.441), 3 (p=.012, ES=0.321), 4 (p=.012, ES=0.340), and 7 (p =.0003, ES=0.514) than in the +3°C group.
Group main effects (CON, +3°C and +6°C) in EMG data (RMS and MF) were presented in Fig. 4A, B. There was intervention effect on RMS (F2,20 =6.614, p =.006) and MF (F2,20 =4.741, p =.021). Passive warming at +6°C resulted in a significant reduction in RMS relative to the CON group (CON 78.6±33.0 vs. +6°C 58.4±22.3%, p =.005, ES=0.717) while no differences were observed between the +3°C and CON groups (CON 78.6±33.0 vs. +3°C 64.8±25.4%, p =.061). MF was higher in the passive warming +6°C group compared to the CON group (CON 81.4±11.0 vs. +6°C 87.5±12.4 Hz, p =.016, ES=0.520) while no differences were observed between the +3°C group compared to the CON group (CON 81.4± 11.0 vs. +3°C 84.0±12.7 Hz, p =.414).
DISCUSSION
This study evaluated the effects of local passive warming administered during the 30 minutes transition periods on subsequent performance of repetitive isokinetic knee extensions. As we hypothesized, the current findings showed that isokinetic exercise performance, measured by total work, peak torque, and average power, was blunted in those who recovered with passive warming (+6°C) (Table 2, Fig. 2A-C). Also, average power was attenuated in the +3°C passive warming group relative to the CON group (Fig. 2C). Thus, the application of a passive warming strategy during the transition period should not be encouraged prior to the performance of isokinetic exercises.
Some previous studies showed that an increased T m improves exercise performance such as. cycling, sprint, or vertical jump [8,12,17,18]. While an increased T m is known to increase muscle contraction speed and rate of force development [15,24,25], changes in muscular force during contraction following elevated T m remained unclear. To answer this, we utilized an isokinetic dynamometer to assess muscular force throughout the range of motion with contraction speed fixed during knee extensions. Current findings suggest that local passive warming prior to exercise might reduce muscle force per se during dynamic muscle contraction (e.g., sprint and vertical jump) while contraction speed and rate of force development for muscle fibers are largely improved, which may mask the decreased muscle force and consequently displays improvement in exercise performance.
Previous findings suggest that passive warming has no benefit and may impair subsequent exercise performance. Cocking et al. [19] showed that passive warming with heated pads at 41.2°C placed on the lower limbs for 20-minute following a 19-minute standardized cycling warm-up provided no additional benefits to repeated sprint performance. Similarly, 30-minute water immersion at 34°C did not improve performance during intense submaximal cycling [31]. Muscular endurance capability during isometric knee extensions were found to be blunted in response to local passive warming intervention [22,23]. Besides, notably similar to our findings, Bailey et al. demonstrated that hot water immersion (HWI) of the lower body at 42°C for 40 minutes diminished the performance of a repetitive knee extensor exercise (time to exhaustion) [21]. They revealed that intramuscular PCr and pH decreased more rapidly in the groups that received a warming treatment, while Pi production was sped up in the heated muscles during exercise [21]. A recent study may explain these results that showed that raising muscle temperature accelerates glycogen synthesis but inhibits glycogen accumulation because of the Pimediated phosphorylase activation [32].
The lower rate of the ATP production via glycolysis required to maintain muscle force may also decrease muscle performance. Edwards et al. observed that after leg immersion at 44°C for 45 minutes, the muscular endurance capacity during repeated isometric contraction was decreased, with only about 22% utilization rate of ATP of total anaerobic sources [22]. Besides, during fatigue protocol, in which the muscle fibers were stimulated to maintain a constant force, the time to peak twitch tension and half-relaxation time decreased when T m was raised, thus the higher frequency of stimulation was needed to maintain the muscular force [22].
An elevation in T m increases the amount of myosin adenosine tri-phosphatase (ATPase) activity [33] and Ca++ sequestration by the sarco-plasmic reticulum [34], resulting in faster muscle contractions and a higher rate of force development [15,24,25]. In the present study, however, muscular contraction speed was fixed at 60°/sec using an isokinetic machine, precluding the possibility that muscle contraction speed influenced muscular performance. The isokinetic exercise performance assessed in the current study was primarily dependent on the ATP-PC and anaerobic glycolytic system. Accordingly, decreases in intramuscular PCr, pH, glycogen accumulation, and the rate of ATP regeneration from anaerobic glycolysis required to maintain the muscle contraction may explain the diminished performance we observed [21,32].
We measured surface electromyography (EMG) from the rectus femoris during the knee extension exercise to assess neuromuscular activity [35]. The root mean square (RMS) of EMG indicates muscle activation during muscle contraction and mean frequency (MF) represents motor neuron firing rate for muscle fibers [36]. In the current study, we observed that EMG RMS was lower in the passive warming (+6°C) than that in the CON group, and MF was higher in the passive warming (+6°C) than that in the CON group and +3°C group following the transition period (Table 3).
This was similar to a study by Thornley et al. [23] who found that the muscular endurance performance was reduced during isometric knee extensions following passive warming. They also revealed that the EMG amplitude decreased, and EMG MF was decreased overtime during each condition; however, MF was higher than that in the control group. The decrease in RMS may refer to the accumulation of metabolites from the ATP hydrolysis, such as H+ or Pi, through small diameter afferents, causing an α-motoneuron inhibition and a reduction supraspinal descending drive [37]. The higher EMG MF was partly due to the increased stimulation frequency required to maintain force output when T m is raised [22]. Overall, EMG analyses in this study showed that local passive warming might decrease muscle activation through inhibition of motor neurons while MF increases to maintain muscular force output.
Considering the trauma associated with direct assessment of muscle temperature, we did not directly measure intramuscular temperature. A previous study done by Faulkner et al. revealed that T m in 3 cm below the muscle fascia of the vastus lateralis increased by 0.4°C following 30 minutes passive warming using heated pants when compared to the control group (no passive warming group) [12].
Given effect sizes observed in group differences, local passive warming administered during the transition period has small to medium effects on reducing isokinetic exercise performance (ES=0.363-0.550) and associated neuromuscular activity (ES=0.232-0.717). Future investigators may need to consider increasing the number of subjects to show obvious effects of such intervention. Based on the T sk data in the current study (Table 1) and methodological similarity of a prior investigation [12], muscle temperature may be elevated in response to local passive warming, although the actual extent is unclear. Moreover, the experiment design of the current investigation did not reflect real sports scenes as there were some unrealistic settings such as exercise type and control group. Future studies are warranted on the causal relationship between the elevated T m and actual sports performance in field.
CONCLUSIONS
The effects of passive warming administered during a 30-minute transition period on muscle strength during isokinetic knee extensions were evaluated. The current study found that local passive warming during the transition period may reduce muscle strength indexed by peak torque, average power, and total work during isokinetic muscle contraction. Although passive warming strategies are widely utilized and there is some evidence to show that it provides some performance benefits for explosive exercise, the application of passive warming should be carefully chosen depending on types of exercise.
Acknowledgements
The experiments were conducted at Jeonbuk National University in Jeonju, South Korea. We extend our gratitude to each of the participants. We would also like to express gratitude to the editors of the Writing Center at Jeonbuk National University for their skilled English language assistance.
Notes
The authors declare that they do not have conflict of interest.
AUTHOR CONTRIBUTION
Conceptualization: X Liu, Y Chang; Data curation: X Liu, Y Chang, SY Lee, JH Lee; Formal analysis: X Liu, Y Chang, SY Lee, JH Lee; Meth-odology: X Liu, Y Chang, SY Lee, JH Lee; Visualization: X Liu, Y Chang; Writing-original draft: X Liu, Y Chang; Writing-review & editing: X Liu, Y Chang.